 You want details of #"atomic radii"# only, not #"ionic radii"#! If you can remember #"Atomic size INCREASES down a Group, but DECREASES"#"across a Period,"# where a Group is a column and Period is a row of the Periodic Table, you have mastered a fundamental principle of chemistry. There is an atomic radius trend on the periodic table of elements where atoms with. The periodic table of elements is organized so that. And note that incomplete valence electronic shells, shield the nuclear charge VERY ineffectively.ĪS a scientist, however, you should seek data that inform your argument. Atomic radius refers to the size of a neutral atom. #Z#, and shielding by other electrons, underlies the structure of the Periodic Table. Electronegativity is the ability of an atom to attract. This contest between nuclear charge, i.e. Therefore, the trend within a group or family on the periodic table is that the atomic size increases with increased number of energy levels. Therefore, the atomic size and atomic radius increase from top to bottom. Elements are grouped according to similar electronic structure, which makes these recurring element properties readily apparent in the periodic table. The bigger the nucleus (the more p+ and n), the stronger the. However, electrons orbit the nucleus rather than cluster together. Their masses contribute to most of the atom's atomic weight because electrons are much much smaller. Atomic radii thus INCREASE down the Group. Use this chart to see at a glance the periodic table trends of electronegativity, ionization energy, atomic radius, metallic character, and electron affinity. The protons and neutrons are clustered tightly in the center of an atom (the nucleus). On the other hand, going down a Group, we go to another so-called shell of electrons, that build on the preceding shell. Describe the trend of atomic radii in the rows in the Periodic Table. 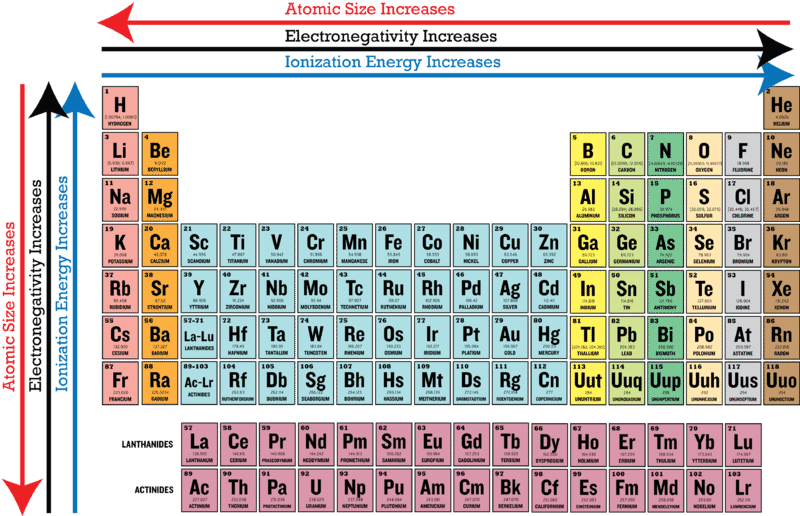
The atomic radii of elements vary in the periodic table in a fixed pattern.  :max_bytes(150000):strip_icc()/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
This results in a DECREASE in atomic radii across the Period, due to the increased nuclear charge which draws in the valence electrons. Describe the general trend in atomic size for groups and for periods. The Atomic radius of an atom is measured by X-ray or other spectroscopy methods. This is because in periods, the valence electrons are in the same outermost shell. In general, the atomic radius decreases as we move from left to right in a period, and it increases when we go down a group. As we go across a Period, a row, of the Periodic Table, from left to right as we FACE the Table, we add another positive charge (a proton, a fundamental, positively charged nuclear particle) to the nucleus. Summary Atomic radius The atomic radius is the distance from the atomic nucleus to the outermost electron orbital in an atom. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
